 These three pieces of data are the elemental symbol, the atomic number (typically given the symbol, Z, and the atomic weight. This periodic table contains all 118 elements along with each element’s atomic number, symbol, name, atomic mass, electron shell configuration, and most common charges (oxidation states). In its simplest form (shown below), each entry only has three pieces of information that you will need to know. This periodic table is a periodic table with charges PDF optimized to fit on a single sheet of paper. You will need to refer to a periodic table for proton values. Use this visual tool for calculating molar mass for any chemical formula. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. The periodic table can often be presented with an abundance of data about each and every element listed. In this notation, the atomic number is not included. Helium is used as a cooling medium for the Large Hadron Collider (LHC), and the superconducting magnets in MRI scanners and NMR spectrometers. A colourless, odourless gas that is totally unreactive. Symbol-mass format for the above atom would be written as Cr-52. Helium was detected in the sun by its spectral lines many years before it was found on Earth. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. Using a periodic table calculate the molar mass of FeSO 4.7H2O What is the percent by mass of water in iron(II) sulfate heptahydrate, FeSO 4.7H 2 O What mass due to waters of crystallization is present in a 4.573g sample of FeSO 4. Usually these instruments only detect hydrogen nuclei in water and fats - ideal for most tissue, but are of no use when looking at air spaces such as the lungs. The "A" value is written as a superscript while the "Z" value is written as a subscript. Xenon-129, a stable isotope that makes up about a quarter of naturally occurring xenon, turns out to be ideal for use in magnetic resonance imaging.  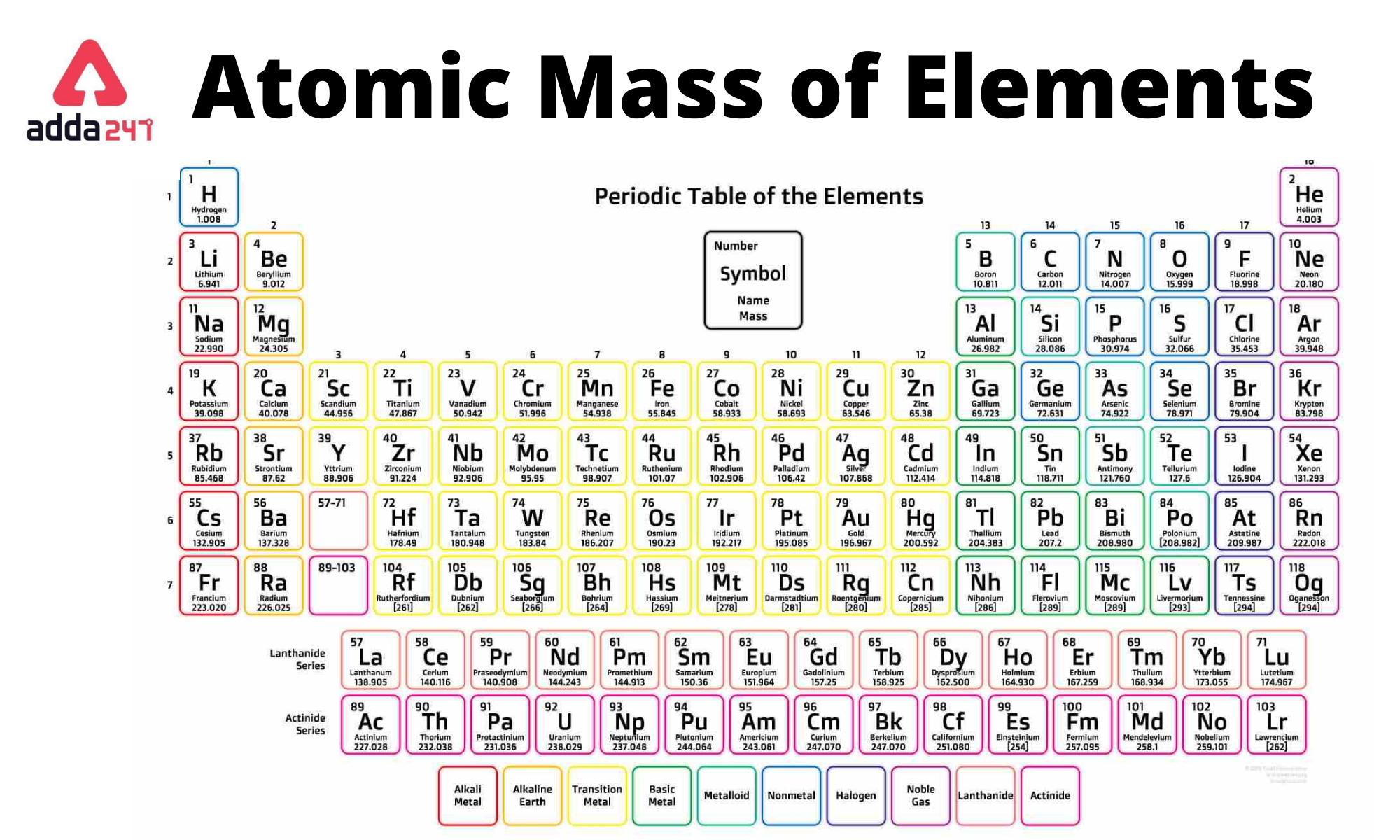
Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
